Mechanism-guided approach to enantiodivergent catalysis
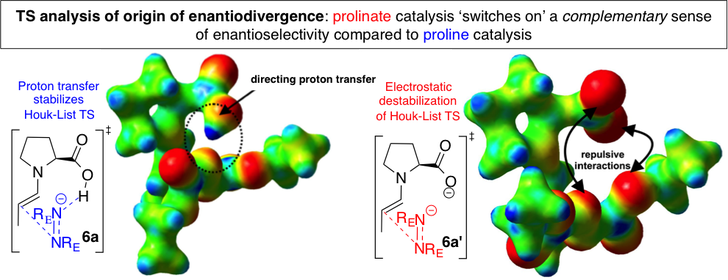
Selective access to either enantiomer of a chiral target molecule using a single chiral catalyst is a very attractive concept to the synthetic chemist, especially when the chiral catalyst is not easily available in both enantiomeric forms. This concept – titled enantiodivergent synthesis – has been explored in a number of specific examples but a general strategy towards realizing this goal is still at large. We attempt to understand the origin of enantiodivergence in already existing examples using transition state analysis and apply the lessons learnt to ‘design’ new enantiodivergent catalytic processes.
Selective access to either enantiomer of a chiral target molecule using a single chiral catalyst is a very attractive concept to the synthetic chemist, especially when the chiral catalyst is not easily available in both enantiomeric forms. This concept – titled enantiodivergent synthesis – has been explored in a number of specific examples but a general strategy towards realizing this goal is still at large. We attempt to understand the origin of enantiodivergence in already existing examples using transition state analysis and apply the lessons learnt to ‘design’ new enantiodivergent catalytic processes.
Identification and application of non-conventional H-bonding interactions in asymmetric organocatalysis
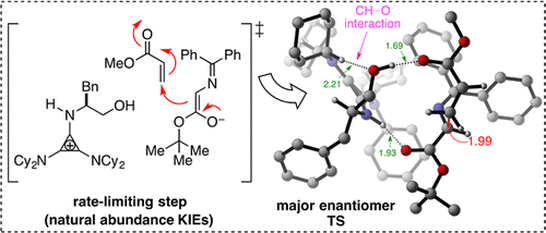
Hydrogen-bonding interactions between polar X-H donors (X = O, N, S) and electronegative acceptor (A) atoms influence reactivity and selectivity in many organic reactions. In contrast, the role of H- bonding interactions between C-H donors and electronegative acceptor atoms has received significantly less attention. Our goal is to obtain high-precision information about how these ‘non- conventional’ CH...A interactions influence transition state assemblies and stereoselectivity in asymmetric organocatalysis. This knowledge is then applied to the rational design of highly selective catalytic processes.
Elucidation of reaction mechanisms in asymmetric organocatalysis
The field of asymmetric organocatalysis has witnessed explosive growth in the past 15 years resulting in hundreds of new catalytic asymmetric reactions. These new transformation can be classified under just a few generic ‘modes’ of substrate activation. Each of these modes of activation encompasses dozens of reactions that can be understood using the same mechanistic framework. Our research program seeks to (a) answers fundamental questions about the broad mechanistic framework that defines each activation mode, and (b) comprehensively investigates fine details of the mechanism of individual reactions within an activation mode that would provide an experimental basis for understanding reactivity and selectivity.