Novel Concepts and Mechanistic Probes in Catalysis: Funded by NIH (R01 GM126283) Dec 2017 -Dec 2022
|
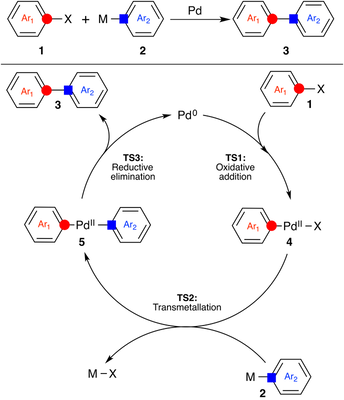
Kinetic Isotope Effects in Palladium Catalyzed Cross-Coupling Reactions Palladium catalyzed cross-coupling (PCC) reactions constitute a very important class of carbon-carbon and carbon-heteroatom bond forming reactions widely used in both academic and industrial settings. This NIH-funded research program utilizes experimental 13C KIEs and density functional theory calculations to address long-standing mechanistic questions in PCC reactions. |
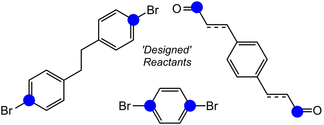
Intramolecular 'Designed' Reactant (IDR) KIE method
Determination of 13C KIEs at natural abundance is a powerful physical organic probe to elucidate fine details of reaction mechanisms. This tool is under-utilized in mechanistic investigations of contemporary catalytic reactions primarily because of large scale required to carry out these experiments. We have developed an innovative yet simple approach that allows for the rapid determination of 13C KIEs using 'designed' reactants - symmetric molecules with two identical reaction sites. The IDR approach can be used for the high-throughput generation of valuable mechanistic information at a fraction of the time and cost of the traditional approaches. We are currently utilizing this approach to study reactions in the area of cross-coupling chemistry, C-H activation chemistry, and all areas of asymmetric organocatalysis.
Determination of 13C KIEs at natural abundance is a powerful physical organic probe to elucidate fine details of reaction mechanisms. This tool is under-utilized in mechanistic investigations of contemporary catalytic reactions primarily because of large scale required to carry out these experiments. We have developed an innovative yet simple approach that allows for the rapid determination of 13C KIEs using 'designed' reactants - symmetric molecules with two identical reaction sites. The IDR approach can be used for the high-throughput generation of valuable mechanistic information at a fraction of the time and cost of the traditional approaches. We are currently utilizing this approach to study reactions in the area of cross-coupling chemistry, C-H activation chemistry, and all areas of asymmetric organocatalysis.
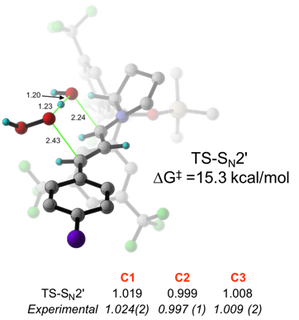
Mechanism-based methodology development in asymmetric organocatalysis
The area of asymmetric organocatalysis has seen explosive growth since the landmark reports by List and Macmillan in 2000. We are experts in the elucidation of complex catalytic cycles in organocatalysis using a combination of experimental and theoretical 13C and 2H KIE studies. Mechanistic studies of enamine catalysis by L-proline conducted in our lab has resulted in resolving a long-standing mechanistic debate in this area. Our studies in the area of iminium catalysis has led to the surprising revelation that the iminium ion can sometimes be an off-cycle intermediate in catalysis. We utilize the mechanistic information from these studies for the rational design and development of new organocatalytic processes. We have active projects in the area of enamine catalysis, iminium catalysis, Brønsted acid and Brønsted base catalysis, H-bonding catalysis by bifunctional ureas/thioureas, and N-heterocyclic carbene catalysis. We are also actively engaged in an increasing number of collaborative mechanistic studies with top methodology groups from around the world.
The area of asymmetric organocatalysis has seen explosive growth since the landmark reports by List and Macmillan in 2000. We are experts in the elucidation of complex catalytic cycles in organocatalysis using a combination of experimental and theoretical 13C and 2H KIE studies. Mechanistic studies of enamine catalysis by L-proline conducted in our lab has resulted in resolving a long-standing mechanistic debate in this area. Our studies in the area of iminium catalysis has led to the surprising revelation that the iminium ion can sometimes be an off-cycle intermediate in catalysis. We utilize the mechanistic information from these studies for the rational design and development of new organocatalytic processes. We have active projects in the area of enamine catalysis, iminium catalysis, Brønsted acid and Brønsted base catalysis, H-bonding catalysis by bifunctional ureas/thioureas, and N-heterocyclic carbene catalysis. We are also actively engaged in an increasing number of collaborative mechanistic studies with top methodology groups from around the world.